|
12/29/2023 0 Comments Compensation flowjo
Solid tissues are the most difficult but single cells can usually be obtained by a combination of mechanical stress, incubation with an enzyme or a combination of both. Lymphoid organs, such as thymus, will usually release single cells after mild mechanical treatment, for example, forcing the tissue through a coarse metal sieve of the type used as a tea strainer. The treatment might alter surface antigens. Cells grown adherent to a culture dish have to be removed from the dish, usually enzymatically. Cells from suspension cultures or from peripheral blood present few problems. Proceed to acquiring the actual staining experiment.The most important feature of sample preparation, as with all samples for flow cytometry, is the production of a suspension of single cells with few clumps and little debris.Repeat Steps 13 and 14 for other tubes, as necessary.Adjust the compensation values until the median fluorescence intensity (MFI) of each population (as shown in the quadrant stats window) is approximately equal). Place a quadrant gate such that the negative bead population is in the lower left quadrant and the positive bead population is in the upper or lower right quadrant.Create a dot plot for the given fluorochrome-conjugated antibody as appropriate.Adjust flow rate to 200–300 events per second if possible.Gate on the singlet bead population based on FSC (forward-light scatter) and SSC (side-light scatter) characteristics. 
Run each tube separately on the flow cytometer.Resuspend the bead pellet in each tube by adding 0.5 mL of staining buffer to each tube.Discard the supernatant from each tube by careful vacuum aspiration using a fine-tip Pasteur pipette.Following the incubation step (see Step 6 above), add 2 mL staining buffer to each tube and pellet by centrifugation at 200 x g for 10 minutes.If you are unsure, use the BD® CompBeads Negative Control Beads as your negative reference point and proceed. During the incubation of beads and antibody, set the flow cytometer instrument PMT voltage settings using the target tissue for the given experiment (e.g., whole blood, splenocytes, etc.).Incubate 15–30 minutes at room temperature.(Make sure the antibody is deposited to the bead mixture, then vortex.) 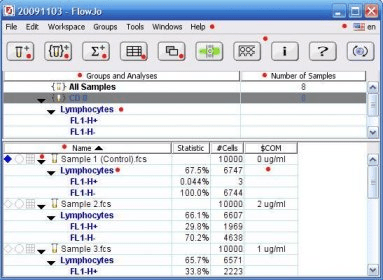
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
